Saturday, June 7, 2008
Sodium Benzoate
Sodium benzoate is a preservative. It is bacteriostatic and fungistatic under acidic conditions. It is used most prevalently in acidic foods such as salad dressings (vinegar), carbonated drinks (carbonic acid), jams and fruit juices (citric acid), pickles (vinegar), and Chinese food sauces (soy, mustard, and duck). It is also found in alcohol-based mouthwash and silver polish. Sodium benzoate is declared on a product label as 'sodium benzoate' or E211. The taste of sodium benzoate cannot be detected by around 25 percent of the population, but for those who can taste the chemical, it tends to be perceived as sweet, sour, salty, or sometimes bitter.
Blended Food Colours
| EGG YELLOW | VC 10 | |
| YOLK YELLOW | VC 11 | |
| ORANGE RED | VC 12 | |
| STRAWBERRY RED | VC 13 | |
| ROSE PINK | VC 14 | |
| RASPBERRY RED | VC 15 | |
| GRAPE | VC 16 | |
| VIOLET | VC 17 | |
| COFFEE BROWN | VC 18 | |
| CHOCOLATE BROWN | VC 19 | |
| DARK CHOCOLATE | VC 20 | |
| LIME GREEN | VC 21 | |
| APPLE GREEN | VC 22 | |
| PEA GREEN | VC 23 | |
| BLACK CURRANT | VC 24 |
| LAKE QUINOLINE YELLOW | 47005:1 | E 104 | |
| LAKE TARTRAZINE | 19140:1 | E 102 | |
| LAKE SUNSET YELLOW FCF | 15985:1 | E 110 | |
| LAKE ERYTHROSINE | 45430:1 | E 127 | |
| LAKE PONCEAU 4R | 16255:1 | E 124 | |
| LAKE ALLURA RED | 16035:1 | E 129 | |
| LAKE CARMOISINE | 14720:1 | E 128 | |
| LAKE AMARANTH | 16185:1 | E 123 | |
| LAKE CHOCCOLATE BROWN HT | 20285:1 | E 155 | |
| LAKE BRILLIANT BLUE FCF | 42090:1 | E 133 | |
| LAKE INDIGO CARMINE | 73015:1 | E 132 |
Synthetic Food Colours

Synthetic food colours, also known as artificial food colours, are manufactured chemically and are the most commonly used dyes in the food, pharmaceutical and cosmetic industries.
| Product/Colour Shade | C.I.No. | F.D. & C.No | E.No. | |
| QUINOLINE YELLOW | 47005 | - | E 104 | |
| ( Di sodium salt of disulfonates of 2-(2quinolyl) - 1, 3 indandione.) | ||||
| TARTRAZINE | 19140 | Yellow 5 | E 102 | |
| ( Tri sodium salt of 5-hydroxy (1-p-sulphophenyl 4- ( p-sulphophenylazo) pyrazol -3- carboxylicacid | ||||
| SUNSET YELLOW FCF | 15985 | Yellow 6 | E 110 | |
| ( Di sodium salt of disulfonates of 2-(2quinolyl) - 1, 3 indandione.) | ||||
| ERYTHROSINE | 45430 | Red 3 | E 127 | |
| ( Di sodium salt of disulfonates of 2-(2quinolyl) - 1, 3 indandione.) | ||||
| PONCEAU 4R | 16255 | - | E 124 | |
| ( Di sodium salt of disulfonates of 2-(2quinolyl) - 1, 3 indandione.) | ||||
| ALLURA RED | 16035 | Red 40 | E 129 | |
| ( Di sodium salt of disulfonates of 2-(2quinolyl) - 1, 3 indandione.) | ||||
| CARMOISINE | 14720 | - | E 122 | |
| ( Di sodium salt of disulfonates of 2-(2quinolyl) - 1, 3 indandione.) | ||||
| AMARANTH | 16185 | Red 2 | E 123 | |
| ( Di sodium salt of disulfonates of 2-(2quinolyl) - 1, 3 indandione.) | ||||
| CHOCOLATE BROWN HT | 20285 | - | E 155 | |
| ( Di sodium salt of disulfonates of 2-(2quinolyl) - 1, 3 indandione.) | ||||
| BRILLIANT BLUE FCF | 42090 | Blue 1 | E 133 | |
| ( Di sodium salt of disulfonates of 2-(2quinolyl) - 1, 3 indandione.) | ||||
| PATENT BLUE V | 42015 | - | E 131 | |
| ( Di sodium salt of disulfonates of 2-(2quinolyl) - 1, 3 indandione.) | ||||
| INDIGO CARMINE | 73015 | Blue 2 | E 132 | |
| ( Di sodium salt of disulfonates of 2-(2quinolyl) - 1, 3 indandione.) | ||||
| BLACK PN | 28440 | - | E 151 | |
| ( Di sodium salt of disulfonates of 2-(2quinolyl) - 1, 3 indandione.) | ||||
| FAST RED E | 16045 | - | - | |
| ( Di sodium salt of disulfonates of 2-(2quinolyl) - 1, 3 indandione.) | ||||
| GREEN S | 44090 | - | E 142 | |
| ( Di sodium salt of disulfonates of 2-(2quinolyl) - 1, 3 indandione.) | ||||
| RED 2G | 18050 | - | E 128 | |
| ( Di sodium salt of 8-actamido-1-hydroxy-2- phenylazonaphthalene-3,6 disulponate) | ||||
Natural Food Colours
 animal, mineral, or source capable of colouring food drug, cosmetic or any part of human body, colours come from variety of sources such as seeds, fruits, vegetables, algae & insect. According to the application a suitable Natural Colour can be achieved by keeping in mind the factors such as PH. heat, light storage and the other ingredients of the formula or recipe. The stirage conditions for natural colours depends on the particular need of the product.
animal, mineral, or source capable of colouring food drug, cosmetic or any part of human body, colours come from variety of sources such as seeds, fruits, vegetables, algae & insect. According to the application a suitable Natural Colour can be achieved by keeping in mind the factors such as PH. heat, light storage and the other ingredients of the formula or recipe. The stirage conditions for natural colours depends on the particular need of the product.A tight sealed container is best to store he product in a cool storage to preserve colour strength and quality, along with its degree of cooling point. r customer specifications.
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Citric Acid

Citric acid is a weak organic acid. It is a natural preservative and is also used to add an acidic, or sour, taste to foods and soft drinks. In biochemistry, it is important as an intermediate in the citric acid cycle and therefore occurs in the metabolism of almost all living things. It also serves as an environmentally benign cleaning agent and acts as an antioxidant.
Citric acid exists in a variety of fruits and vegetables, most notably citrus fruits. Lemons and limes have particularly high concentrations of the acid; it can comprise as much as 8% of the dry weight of these fruits (1.44 and 1.38 grams per ounce of the juices, respectively).
Citric acid has been used as an additive to soft drinks, beer, and seltzer, and occurs naturally in many juices. This causes a problem in measurement because the standard measuring technique for sugar is refractive index. The refractive index of sugar and citric acid is almost identical. For soft drinks and orange juice the best measure of sweetness is the sugar/acid ratio. Recently, the use of infrared sensors has allowed measurement of both Brix (sugar content) and acidity by detecting sugars and citric acid through their characteristic molecular vibrations; this gives an accurate assessment of a drink's sweetness.
As a food additive, citric acid is used as a flavouring and preservative in food and beverages, especially soft drinks. It is denoted by E number E330. Citrate salts of various metals are used to deliver those minerals in a biologically available form in many dietary supplements. The buffering properties of citrates are used to control pH in household cleaners and pharmaceuticals.
Flavour

Flavor or flavour is the sensory impression of a food or other substance, and is determined mainly by the chemical senses of taste and smell. The "trigeminal senses", which detect chemical irritants in the mouth and throat, may also occasionally determine flavor. The flavor of the food, as such, can be altered with natural or artificial flavorants, which affect these senses.
Flavorant is defined as a substance that gives another substance flavor, altering the characteristics of the solute, causing it to become sweet, sour, tangy, etc.
Of the three chemical senses, smell is the main determinant of a food item's flavor. While the taste of food is limited to sweet, sour, bitter, salty, and savory (umami)--the basic tastes--the smells of a food are potentially limitless. A food's flavor, therefore, can be easily altered by changing its smell while keeping its taste similar. Nowhere is this better exemplified than in artificially flavored jellies, soft drinks and candies, which, while made of bases with a similar taste, have dramatically different flavors due to the use of different scents or fragrances.
Although the terms "flavoring" or "flavorant" in common language denote the combined chemical sensations of taste and smell, the same terms are usually used in the fragrance and flavors industry to refer to edible chemicals and extracts that alter the flavor of food and food products through the sense of smell. Due to the high cost or unavailability of natural flavor extracts, most commercial flavorants are nature-identical, which means that they are the chemical equivalent of natural flavors but chemically synthesized rather than being extracted from the source materials.
- Natural flavouring substances: Flavouring substances obtained from plant or animal raw materials, by physical, microbiological or enzymatic processes. They can be either used in their natural state or processed for human consumption, but cannot contain any nature-identical or artificial flavouring substances.
- Nature-identical flavouring substances: Flavouring substances that are obtained by synthesis or isolated through chemical processes, which are chemically identical to flavouring substances naturally present in products intended for human consumption. They cannot contain any artificial flavouring substances.
- Artificial flavouring substances: Flavouring substances not identified in a natural product intended for human consumption, whether or not the product is processed.
Most food and beverage companies do not create their own flavors but instead employ the services of a flavour company. Food and beverage companies may require flavors for new products, product line extensions (e.g., low fat versions of existing products) or due to changes in formula or processing for existing products.
The flavor creation is done by a specially trained scientist called a "Flavourist". Flavourist's job combines extensive scientific knowledge of the chemical palette wit$h artistic creativity to develop new and distinctive flavors. The flavor creation begins when the flavorist receives a brief from the client. In the brief the client will attempt to communicate exactly what type of flavor they seek, in what application it will be used, and any special requirements (e.g., must be all natural). The communication barrier can be quite difficult to overcome since most people aren't experienced at describing flavors. The flavorist will use his or her knowledge of the available chemical ingredients to create a formula and compound it on an electronic balance. The flavor will then be submitted to the client for testing. Several iterations, with feedback from the client, may be needed before the right flavor is found.
Additional work may also be done by the flavor company. For example, the flavor company may conduct sensory taste tests to test consumer acceptance of a flavor before it is sent to the client or to further investigate the "sensory space." The flavor company may also employ application specialists who work to ensure the flavor will work in the application for which it is intended. This may require special flavor delivery technologies that are used to protect the flavor during processing or cooking so that the flavor is only released when eaten by the end consumer.
Sunday, June 1, 2008
Health effects of drinking Soda
Intake of carbonated beverages has not been associated with increased bone fracture risk in observational studies, and the net effect of carbonated beverage constituents on the amount of calcium in the body is negligible, leaving carbonated water as harmless as regular water.
History of Carbonated water (Soda)
In 1771 Swedish chemistry professor Torbern Bergman independently invented a similar process to make carbonated water. In poor health at the time yet frugal, he was trying to reproduce naturally-effervescent spring waters thought at the time to be beneficial to health.
Today, carbonated water is made by passing pressurized carbon dioxide through water. The pressure increases the solubility and allows more carbon dioxide to dissolve than would be possible under standard atmospheric pressure. When the bottle is opened, the pressure is released, allowing the gas to come out of the solution, thus forming the characteristic bubbles.
In the US, Carbonated water was commonly known by the name of soda water until World War II. In the 1950's new terms such as sparkling water and seltzer water began to be used. The term seltzer water is a genericized trademark that comes from the German brand Selters, which is produced and bottled in Nieder-Selters, Germany.
Flavored carbonated water is also commercially available. It differs from sodas in that it contains flavors (usually fruit flavors such as lemon, lime, orange, or raspberry) but no sugar.
Carbonated Water
 In the past, soda water was produced in the home by "charging" a refillable seltzer bottle by filling it with water and then adding carbon dioxide. Club soda may be identical to plain carbonated water or it may contain a small amount of table salt, sodium citrate, sodium bicarbonate, potassium bicarbonate, potassium sulfate, or disodium phosphate, depending on the bottler. These additives are included to emulate the slightly salty taste of homemade soda water. The process can also occur naturally to produce carbonated mineral water.
In the past, soda water was produced in the home by "charging" a refillable seltzer bottle by filling it with water and then adding carbon dioxide. Club soda may be identical to plain carbonated water or it may contain a small amount of table salt, sodium citrate, sodium bicarbonate, potassium bicarbonate, potassium sulfate, or disodium phosphate, depending on the bottler. These additives are included to emulate the slightly salty taste of homemade soda water. The process can also occur naturally to produce carbonated mineral water.
Uses of Carbon dioxide
Carbon dioxide is used by the food industry, the oil industry, and the chemical industry. It is used in many consumer products that require pressurized gas because it is inexpensive and nonflammable, and because it undergoes a phase transition from gas to liquid at room temperature at an attainable pressure of approximately 60 bar (870 psi, 59 atm), allowing far more carbon dioxide to fit in a given container than otherwise would. Life jackets often contain canisters of pressured carbon dioxide for quick inflation. Aluminum capsules are also sold as supplies of compressed gas for airguns, paintball markers, for inflating bicycle tires, and for making seltzer. Rapid vaporization of liquid carbon dioxide is used for blasting in coal mines. High concentrations of carbon dioxide can also be used to kill pests, such as the Common Clothes Moth.
Leavening agents produce carbon dioxide to cause dough to rise. Baker's yeast produces carbon dioxide by fermentation of sugars within the dough, while chemical leaveners such as baking powder and baking soda release carbon dioxide when heated or if exposed to acids.
A carbon dioxide laser.Carbon dioxide is the most commonly used compressed gas for pneumatic systems in portable pressure tools and combat robots.
Carbon dioxide extinguishes flames, and some fire extinguishers, especially those designed for electrical fires, contain liquid carbon dioxide under pressure. Carbon dioxide also finds use as an atmosphere for welding, although in the welding arc, it reacts to oxidize most metals. Use in the automotive industry is common despite significant evidence that welds made in carbon dioxide are brittler than those made in more inert atmospheres, and that such weld joints deteriorate over time because of the formation of carbonic acid. It is used as a welding gas primarily because it is much less expensive than more inert gases such as argon or helium.
Liquid carbon dioxide is a good solvent for many lipophilic organic compounds, and is used to remove caffeine from coffee. First, the green coffee beans are soaked in water. The beans are placed in the top of a column seventy feet (21 meters) high. The carbon dioxide fluid at about 93 degrees Celsius enters at the bottom of the column. The caffeine diffuses out of the beans and into the carbon dioxide.
Carbon dioxide has begun to attract attention in the pharmaceutical and other chemical processing industries as a less toxic alternative to more traditional solvents such as organochlorides. It's used by some dry cleaners for this reason.
Plants require carbon dioxide to conduct photosynthesis, and greenhouses may enrich their atmospheres with additional CO2 to boost plant growth, since its low present-day atmosphere concentration is just above the "suffocation" level for green plants. A photosynthesis-related drop in carbon dioxide concentration in a greenhouse compartment can kill green plants. At high concentrations, carbon dioxide is toxic to animal life, so raising the concentration to 10,000 ppm (1%) for several hours can eliminate pests such as whiteflies and spider mites in a greenhouse.
It has been proposed that carbon dioxide from power generation be bubbled into ponds to grow algae that could then be converted into biodiesel fuel. In medicine, up to 5% carbon dioxide is added to pure oxygen for stimulation of breathing after apnea and to stabilize the O2/CO2 balance in blood.
A common type of industrial gas laser is the carbon dioxide laser.
Carbon dioxide can also be combined with limonene oxide from orange peels or other epoxides to create polymers and plastics.
Carbon dioxide is used in enhanced oil recovery where it is injected into or adjacent to producing oil wells, usually under supercritical conditions. It acts as both a pressurizing agent and, when dissolved into the underground crude oil, significantly reduces its viscosity, enabling the oil to flow more rapidly through the earth to the removal well. In mature oil fields, extensive pipe networks are used to carry the carbon dioxide to the injection points.
In the chemical industry, carbon dioxide is used for the production of urea, carbonates and bicarbonates, and sodium salicylate.
Liquid and solid carbon dioxide are important refrigerants, especially in the food industry, where they are employed during the transportation and storage of ice cream and other frozen foods. Solid carbon dioxide is called "dry ice" and is used for small shipments where refrigeration equipment is not practical.
Liquid carbon dioxide (industry nomenclature R744 / R-744) was used as a refrigerant prior to the discovery of R-12 and is likely to enjoy a renaissance due to environmental concerns. Its physical properties are highly favorable for cooling, refrigeration, and heating purposes, having a high volumetric cooling capacity. Due to its operation at pressures of up to 130 bars, CO2 systems require highly resistant components that have been already developed to serial production in many sectors. In car air conditioning, in more than 90% of all driving conditions, R744 operates more efficiently than systems using R-134a. Its environmental advantages (GWP of 1, non-ozone depleting, non-toxic, non-flammable) could make it the future working fluid to replace current HFCs in cars, supermarkets, hot water heat pumps, among others. Some applications: Coca-Cola has fielded CO2-based beverage coolers and the US Army is interested in CO2 refrigeration and heating technology.
By the end of 2007, the global car industry is expected to decide on the next-generation refrigerant in car air conditioning. CO2 is one discussed option.
Enhanced Coal Bed Methane recovery (ECBM) is the process whereby carbon dioxide is pumped into the coal seam to displace methane thanks to higher CO2 adsorptivity.
Toxicity of Carbon dioxide
A person's exhaled breath is approximately 4.5% carbon dioxide by volume.
Adaptation to increased levels of CO2 occurs in humans. Continuous inhalation of CO2 can be tolerated at three percent inspired concentrations for at least one month and four percent inspired concentrations for over a week. It was suggested that 2.0 percent inspired concentrations could be used for closed air spaces (ex. Submarine) since the adaptation is physiological and reversible. Decrement in performance or in normal physical activity does not happen at this level.
It is dangerous when inhaled in high concentrations (greater than 5% by volume, or 50,000 ppm). The current threshold limit value (TLV) or maximum level that is considered safe for healthy adults for an eight-hour work day is 0.5% (5,000 ppm). The maximum safe level for infants, children, the elderly and individuals with cardio-pulmonary health issues is significantly less.
These figures are valid for pure carbon dioxide. In indoor spaces occupied by people the carbon dioxide concentration will reach higher levels than in pure outdoor air. Concentrations higher than 1,000 ppm will cause discomfort in more than 20% of occupants, and the discomfort will increase with increasing CO2 concentration. The discomfort will be caused by various gases coming from human respiration and perspiration, and not by CO2 itself. At 2,000 ppm the majority of occupants will feel a significant degree of discomfort, and many will develop nausea and headaches. The CO2 concentration between 300 and 2,500 ppm is used as an indicator of indoor air quality.
Acute carbon dioxide toxicity is sometimes known as by the names given to it by miners: blackdamp (also called choke damp or stythe). Miners would try to alert themselves to dangerous levels of carbon dioxide in a mine shaft by bringing a caged canary with them as they worked. The canary would inevitably die before CO2 reached levels toxic to people. Carbon dioxide caused a great loss of life at Lake Nyos in Cameroon in 1986, when an upwelling of CO2-laden lake water quickly blanketed a large surrounding populated area. The heavier carbon dioxide forced out the life-sustaining oxygen near the surface, killing nearly two thousand people.
Carbon dioxide ppm levels (CDPL) are a surrogate for measuring indoor pollutants that may cause occupants to grow drowsy, get headaches, or function at lower activity levels. To eliminate most Indoor Air Quality complaints, total indoor CDPL must be reduced to below 600. NIOSH considers that indoor air concentrations that exceed 1,000 are a marker suggesting inadequate ventilation. ASHRAE recommends they not exceed 1,000 inside a space. OSHA limits concentrations in the workplace to 5,000 for prolonged periods. The U.S. National Institute for Occupational Safety and Health limits brief exposures (up to ten minutes) to 30,000 and considers CDPL exceeding 40,000 as "immediately dangerous to life and health." People who breathe 50,000 for more than half an hour show signs of acute hypercapnia, while breathing 70,000 – 100,000 can produce unconsciousness in only a few minutes. Accordingly, carbon dioxide, either as a gas or as dry ice, should be handled only in well-ventilated areas.
Carbon dioxide Manufacture
1. As a byproduct in ammonia and hydrogen plants, where methane is converted to CO2;
2. From combustion of wood and fossil fuels;
3. As a byproduct of fermentation of sugar in the brewing of beer, whisky and other alcoholic beverages;
4. From thermal decomposition of limestone, CaCO3, in the manufacture of lime, CaO;
5. As a byproduct of sodium phosphate manufacture;
6. Directly from natural carbon dioxide springs, where it is produced by the action of acidified water on limestone or dolomite.
Isolation of Carbon dioxide
H2SO4 + CaCO3 → CaSO4 + H2CO3
The H2CO3 then decomposes to water and CO2. Such reactions are accompanied by foaming or bubbling, or both. In industry such reactions are widespread because they can be used to neutralize waste acid streams.
The production of quicklime (CaO) a chemical that has widespread use, from limestone by heating at about 850 °C also produces CO2:
CaCO3 → CaO + CO2
The combustion of all carbon containing fuels, such as methane (natural gas), petroleum distillates (gasoline, diesel, kerosene, propane), but also of coal and wood, will yield carbon dioxide and, in most cases, water. As an example the chemical reaction between methane and oxygen is given below.
CH4 + 2 O2 → CO2 + 2 H2O
Iron is reduced from its oxides with coke in a blast furnace, producing pig iron and carbon dioxide:
2 Fe2O3 + 3 C → 4 Fe + 3 CO2
Yeast metabolizes sugar to produce carbon dioxide and ethanol, also known as alcohol, in the production of wines, beers and other spirits:
C6H12O6 → 2 CO2 + 2 C2H5OH
All aerobic organisms produce CO2 when they oxidize carbohydrates, fatty acids, and proteins in the mitochondria of cells. The large number of reactions involved are exceedingly complex and not described easily. Refer to (cellular respiration, anaerobic respiration and photosynthesis). Photoautotrophs (i.e. plants, cyanobacteria) use another modus operandi: Plants absorb CO2 from the air, and, together with water, react it to form carbohydrates:
nCO2 + nH2O → (CH2O)n + nO2
Carbon dioxide is soluble in water, in which it spontaneously interconverts between CO2 and H2CO3 (carbonic acid). The relative concentrations of CO2, H2CO3, and the deprotonated forms HCO3− (bicarbonate) and CO32−(carbonate) depend on the pH. In neutral or slightly alkaline water (pH > 6.5), the bicarbonate form predominates (>50%) becoming the most prevalent (>95%) at the pH of seawater, while in very alkaline water (pH > 10.4) the predominant (>50%) form is carbonate. The bicarbonate and carbonate forms are very soluble, such that air-equilibrated ocean water (mildly alkaline with typical pH = 8.2 – 8.5) contains about 120 mg of bicarbonate per liter.
History of Carbon dioxide
The properties of carbon dioxide were studied more thoroughly in the 1750s by the Scottish physician Joseph Black. He found that limestone (calcium carbonate) could be heated or treated with acids to yield a gas he called "fixed air." He observed that the fixed air was denser than air and did not support either flame or animal life. He also found that when bubbled through an aqueous solution of lime (calcium hydroxide), it would precipitate calcium carbonate. He used this phenomenon to illustrate that carbon dioxide is produced by animal respiration and microbial fermentation. In 1772, English chemist Joseph Priestley published a paper entitled Impregnating Water with Fixed Air in which he described a process of dripping sulfuric acid (or oil of vitriol as Priestley knew it) on chalk in order to produce carbon dioxide, and forcing the gas to dissolve by agitating a bowl of water in contact with the gas.
Carbon dioxide was first liquefied (at elevated pressures) in 1823 by Humphry Davy and Michael Faraday. The earliest description of solid carbon dioxide was given by Charles Thilorier, who in 1834 opened a pressurized container of liquid carbon dioxide, only to find that the cooling produced by the rapid evaporation of the liquid yielded a "snow" of solid CO2.
Chemical & Physical properties of Carbon dioxide

Carbon dioxide is a colorless, odorless gas. When inhaled at concentrations much higher than usual atmospheric levels, it can produce a sour taste in the mouth and a stinging sensation in the nose and throat. These effects result from the gas dissolving in the mucous membranes and saliva, forming a weak solution of carbonic acid. This sensation can also occur during an attempt to stifle a burp after drinking a carbonated beverage. Amounts above 5,000 ppm are considered very unhealthy, and those above about 50,000 ppm (equal to 5% by volume) are considered dangerous to animal life.
At standard temperature and pressure, the density of carbon dioxide is around 1.98 kg/m³, about 1.5 times that of air. The carbon dioxide molecule (O=C=O) contains two double bonds and has a linear shape. It has no electrical dipole, and as it is fully oxidized, it is moderately reactive and is non-flammable, but will support the combustion of metals such as magnesium.
At −78.51° C or -109.3° F, carbon dioxide changes directly from a solid phase to a gaseous phase through sublimation, or from gaseous to solid through deposition. Solid carbon dioxide is normally called "dry ice", a generic trademark. It was first observed in 1825 by the French chemist Charles Thilorier. Dry ice is commonly used as a cooling agent, and it is relatively inexpensive. A convenient property for this purpose is that solid carbon dioxide sublimes directly into the gas phase leaving no liquid. It can often be found in grocery stores and laboratories, and it is also used in the shipping industry. The largest non-cooling use for dry ice is blast cleaning.
Liquid carbon dioxide forms only at pressures above 5.1 atm; the triple point of carbon dioxide is about 518 kPa at −56.6 °C (See phase diagram, above). The critical point is 7.38 MPa at 31.1 °C.[3]
An alternative form of solid carbon dioxide, an amorphous glass-like form, is possible, although not at atmospheric pressure.[4] This form of glass, called carbonia, was produced by supercooling heated CO2 at extreme pressure (40–48 GPa or about 400,000 atmospheres) in a diamond anvil. This discovery confirmed the theory that carbon dioxide could exist in a glass state similar to other members of its elemental family, like silicon (silica glass) and germanium. Unlike silica and germania glasses, however, carbonia glass is not stable at normal pressures and reverts back to gas when pressure is released.
Carbon dioxide

Carbon dioxide is produced by all animals, plants, fungi and microorganisms during respiration and is used by plants during photosynthesis. This is to make sugars which may either be consumed again in respiration or used as the raw material for plant growth. It is, therefore, a major component of the carbon cycle. Carbon dioxide is generated as a byproduct of the combustion of fossil fuels or vegetable matter, among other chemical processes. Inorganic carbon dioxide is output by volcanoes and other geothermal processes such as hot springs.
Carbon dioxide has no liquid state at pressures below 5.1 atm, but is a solid at temperatures below -78 °C. In its solid state, carbon dioxide is commonly called dry ice.
What is a Softdrink
Uses of Carbonation
Natural and artificial carbonation
How to measure Carbonation ?
Foaming of Carbonated Drinks
It is commonly said that shaking a carbonated beverage will cause large amounts of foam to erupt upon opening, and it is often believed that shaking a bottle containing a carbonated beverage will cause the pressure inside to rise. Contrary to this myth, when a pressure gauge is attached to a pressurized bottle of a carbonated beverage, it is found that the pressure within does not increase. It is instead the formation of tiny bubbles from the agitation that causes the foam. Upon opening, the size of the bubbles will rapidly increase due to the reduction in pressure, resulting in excessive foaming.
Agitating carbonated liquid in a resealed vessel increases the rate that CO2 is released from the solution and the rate that it approaches equilibrium pressure. The equilibrium pressure depends on the temperature, composition, and purity of the liquid, not the agitation of it.
Fizzle !
A carbonated beverage, such as cola or beer, will form bubbles when the dissolved carbon dioxide is depressurized to form emulsions at the top, and it will make "fizzing" sounds when it is opened or poured into a container. In the United Kingdom, soft drinks are often referred to as 'fizzy drinks'. A cocktail based on carbonated water and an acidic juice is called a Fizz, such as the Gin Fizz.
What is Effervescence?
This process is generally represented by the following reaction, where a pressurized dilute solution of carbonic acid in water releases gaseous carbon dioxide at decompression:
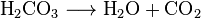
In simple terms, it is the result of the chemical reaction occurring in the liquid which produces a gaseous product.
What is Carbonation ?

There are several definitions for Carbonation
1. Carbonation occurs when carbon dioxide is dissolved in water or an aqueous solution. This process yields the "fizzle" to carbonated water and sparkling mineral water, the head to beer, and the cork pop and bubbles to champagne and sparkling wine.
2. Sparkle caused by carbon dioxide, either created during fermentation or injected later.
3. This term is used to describe both the amount of CO2 in the beverage, as well as the process of putting CO2 into the beverage. There are two types of carbonation - forced carbonation and natural carbonation
Introduction
With over 10 years of experience in Food & Beverages Industry and into Fizzy (carbonated ) drinks manufacture, quality control, operation & maintenance of Carbonation & filling equipment, I now want to start off with this blog of Interesting things I experience day to day which is related to Carbonation .
If you find anything inapropriate, would like to comment or would like to contribute more to the development of this blog, plz dont hesitate to email me > sridharwins@gmail.com
Enjoy the blog!
Regards,
Sridhar
31-MAY-2008 22:14 hrs






